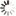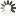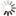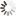| brand Johnson & | Johnson |
|---|
| model | Johnson & Johnson Day Throw 30 Tablets |
|---|
| Material | Hydrogel |
|---|
| Packaging Type | Approval Document Number for Ordinary Packaging |
|---|
| National Machinery Injection 20193162188 |
|---|
| Lens Classification | Myopia |
|---|
| Wearing Cycle | daily abandoned |
|---|
| production enterprise | johnsonjohnson vision care in |
|---|
| Irish degree of origin | |
|---|
| 100 125 150 175 200 225 250 275 300 325 350 375 400 425 450 |
|---|
| Trade name of contact lenses and treatment fluids | Johnson & Johnson Day Throw 30 Tablets |
|---|
| Manufacturer's address | 7500 Centurion Parkway Jackson |
|---|
| Packaging size | 30 tablets |
|---|
| lens oxygen permeability | 191 x 10 Å (cm s) (mLO ₂ mLxhPa)] |
|---|
| radius of base arc | 85mm |
|---|
| UV absorption | Category 2 |
|---|
| water content | 59% |
|---|
| Center thickness | 0084mm |
|---|
| Total diameter | 142mm |
|---|


















![Contains mirror box] Haichang Nursing Lotion Sensation 120ml * 4 Portable Vials Contact Lens Myopia Eyewear Flushing Lotion](http://img.alicdn.com/imgextra/i4/0/O1CN01C7XmJX1ahqMa5syug_!!0-item_pic.jpg_210x210.jpg)